
Designed by Paul Smith 2006. This website is copyrighted by law.
Material contained herewith may not be used without the prior written permission of FAUNA Paraguay.
Photographs on this web-site were taken by Paul Smith, Hemme Batjes, Regis Nossent, Frank Fragano,
Alberto Esquivel, Arne Lesterhuis, José Luis Cartes, Rebecca Zarza and Hugo del Castillo and are used with their permission.
Phyllostomus discolor (JA Wagner 1843)
TAXONOMY: Class Mammalia; Subclass Theria; Infraclass Metatheria; Order Chiroptera; Suborder Microchiroptera; Superfamily Noctilionoidea; Family Phyllostomidae, Subfamily Phyllostominae (López-Gonzalez 2005, Myers et al 2006). There are four species in this genus, two of which occur in Paraguay. The generic name Phyllostomus is Greek meaning leaf mouth in relation to the prominent nose leaf. The species name discolor is Latin meaning variegated or of different colours in relation to the variably tricoloured hairs of the dorsal pelage which give a variegated appearance (Braun & Mares 1995).
The type specimen is an adult male from Cuyabá (=Cuiabá), Brazil, deposited in the Zoologisches Staats-Sammlung, Munich, Germany. Despite the extensive geographic range the species was considered variable but monotypic by Power & Tamsitt (1973) following a morphological review of the specimens. They noted patterns of pronounced heterogeneity in populations west of the Andes and into Central America, with populations east of the Andes more homogeneous. Valdez (1970) had earlier recognised P.d.verrucosum Elliot, 1905 for the variable Central American and west of the Andes populations, but Power & Tamsitt (1973) noted that populations close to the Andes and on other side exhibit intermediate characters. Willig (1983) found significant differences in cranial and external measurements for populations from northeastern Brazil. A comprehensive modern analysis is required to fully understand the complex geographical variation in this variable species and for now we prefer to follow Gardner (2007) in considering the species monotypic. Synonyms adapted from Gardner (2007) and López-González (2005):
Phyllostoma discolor JA Wagner 1843:366. Type locality "Cuyaba" Mato Grosso, Brazil.
Ph[yllostoma]. innominatum Tschudi 1844:62. Type locality "Maynas" Loreto, Peru.
Phyllostoma angusticeps P.Gervais 1856:47 Type locality "Provincia de Bahía" Brazil.
Phyllostomus discolor JA Allen 1904:344. First use of current name combination.
Phyllostoma verrucossum Elliot 1905:236 Type locality "Niltepec, Oaxaca, Mexico".
P[hyllostomus] verrucosus Miller 1907:131. Name combination and emendation.
ENGLISH COMMON NAMES: Pale Spear-nosed Bat (Gardner 2007, Redford & Eisenberg 1992), Lesser Spear-nosed Bat (Goodwin & Greenhall 1961), Long-tongued Spear-nosed Bat (Goodwin & Greenhall 1961).
SPANISH COMMON NAMES: Falso vampiro gris (Redford & Eisenberg 1992), Murciélago nariz de lanza pálido (Aguirre 2007), Murciélago que hoja nasal lanceolada (Emmons 1999).
GUARANÍ COMMON NAMES: No known names.
DESCRIPTION: A robust, medium-sized bat with a well-developed nose leaf. Pelage smooth and dense, not extending to the membranes. Overall colour brownish with dorsal surface having somewhat grizzled appearance due to tricoloured hairs - basally white, broad central band dark brown and tip ashy. Head and shoulders slightly paler and underparts notably paler, varying from cinnamon-buff to creamy-white. Ears narrow and somewhat pointed, well-separated on head. Lower lip with V-shaped groove bordered by elongate papillae. Uropatagium extensive.
CRANIAL CHARACTERISTICS: Skull robust but long and slender whencompared to other Phyllostomids. Rostrum broad and low, braincase rounded and sagittal crest poorly developed. Goodwin & Greenhall (1981).
Power & Tamsitt (1973) found a significant difference in measurements of mastoid width and zygomatic width between sexes with males being larger. They noted the following mean measurements from across the species wide range (n=138 males, 85 females): Greatest Skull Length male 30.39mm, female 29.61mm; Interorbital Constriction male 6.55mm, female 6.43mm; Condyloincisive Length male 27.33mm, female 26.67mm; Zygomatic Width male 15.65mm, female 15.21mm; Mastoid Width male 14.81mm, female 14.25mm; Width of Braincase male 12.19mm, female 11.97mm; Width Across Molars male 10.11mm, female 9.97mm.
Valdivieso & Tamsitt (1962) give the following ranges of measurements for two skulls from Colombia: Greatest Skull Length 29.8mm 31mm; Interorbital Constriction 6.5mm 6.5mm; Length of Mandible 20.2mm 20.5mm; Zygomatic Width 14.5mm 15.4mm; Mastoid Width 14.1mm 14.7mm; Width Across Molars 9.9mm 10.2mm. Anderson (1997) gives the following ranges for 4 males from Bolivia: Condylobasal Length 26.7-27.2mm; Zygomatic Width 15.1-16mm; Lamboidal Width 14.4-15.4mm; Width of Braincase 11.6-12.2mm; Skull Depth 10.9-11-7mm; Width Across Canines 6.8-7.5mm.
Taddei (1975) gives the following measurements for a sexed sample (n=15 males, n=15 females) from northeast Brazil: Greatest Skull Length male 29.50mm (+/- 0.15mm) female 29.27mm (+/- 0.12mm); Condylobasal Length male 26.61mm (+/- 0.13mm) female 26.30mm (+/- 0.12mm); Condylocaninal Length male 25.35mm (+/- 0.15mm) female 25.17mm (+/- 0.11mm); Basal Skull Length male 22.63mm (+/- 0.12mm) female 22.57mm (+/- 0.11mm); Length of Palate male 12.66mm (+/- 0.06mm) female 12.57mm (+/- 0.08mm); Length of Mandible male 19.61mm (+/- 0.13mm) female 19.50mm (+/- 0.09mm); Width Across Upper Canines male 7.34mm (+/- 0.04mm) female 6.91mm (+/- 0.05mm); Width Across Upper Molars male 10.10mm (+/- 0.04mm) female 9.83mm (+/- 0.08mm); Postorbital Width male 6.55mm (+/- 0.07mm) female 6.43mm (+/- 0.05mm); Transverse Zygomatic Width male 15.55mm (+/- 0.06mm) female 15.55mm (+/- 0.12mm); Width of Braincase male 12.25mm (+/- 0.06mm) female 12.24mm (+/- 0.06mm); Mastoid Width male 14.90mm (+/- 0.06mm) female 14.62mm (+/- 0.06mm); Height of Braincase male 10.95mm (+/- 0.03mm) female 10.76mm (+/- 0.06mm).
DENTAL CHARACTERISTICS: I2/2 C 1/1 P2/2 M3/3 = 32. Outer incisors short and broad with lower incisors forming a continuous row between the canines and outer tooth only slightly smaller than inner. Cutting edges faintly trifid. Goodwin & Greenhall (1981).
Valdivieso & Tamsitt (1962) give the following measurement for two skulls from Colombia: Upper Tooth Row 9mm. Anderson (1997) gives the following ranges for 4 males from Bolivia: Molar Width 2.1-2.2mm; Dental Span 9.5-10mm. Power & Tamsitt (1973) gave the following mean measurements from across the species wide range (n=138 males, 85 females): Upper Tooth Row male 9.75mm, female 9.57mm.
Taddei (1975) gives the following measurements for a sexed sample (n=30 males, n=30 females) from northeast Brazil: HB: male 79.99mm (+/- 0.42mm) female 80.62mm (+/- 0.45mm); FT male 12.72mm (+/- 0.09mm) female 10.88mm (+/- 0.11mm); FA male 62.50mm (+/- 0.32mm) female 62.68mm (+/- 0.27mm); EA male 21.65mm (+/- 0.12mm) female 22.52mm (+/- 0.15mm); Height of tragus male 6.45mm (+/- 0.07mm) female 6.50mm (+/- 0.06mm); Third Metacarpal male 54.30mm (+/- 0.30mm) female 54.37mm (+/- 0.32mm); First Phalange of Third Digit male 14.50mm (+/- 0.13mm) female 14.75mm (+/- 0.13mm); Second Phalange of Third Digit male 26.67mm (+/- 0.22mm) female 27.06mm (+/- 0.22mm); Third Phalange of Third Digit male 13.82mm (+/- 0.21mm) female 14.22mm (+/- 0.17mm); Fourth Metacarpal male 53.16mm (+/- 0.27mm) female 53.25mm (+/- 0.31mm); First Phalange of Fourth Digit male 11.32mm (+/- 0.11mm) female 11.35mm (+/- 0.10mm); Second Phalange of Fourth Digit male 18.00mm (+/- 0.14mm) female 18.35mm (+/- 0.15mm); Fifth Metacarpal male 52.75mm (+/- 0.32mm) female 53.13mm (+/- 0.26mm); First Phalange of Fifth Digit male 9.97mm (+/- 0.06mm) female 10.13mm (+/- 0.11mm); Second Phalange of Fifth Digit male 12.47mm (+/- 0.12mm) female 13.03mm (+/- 0.14mm); Tibia male 23.75mm (+/- 0.19mm) female 23.92mm (+/- 0.18mm); Calcar male 10.83mm (+/- 0.11mm) female 10.88mm (+/- 0.11mm); WT: male 37.18g (+/- 0.55g, n=16), subadult males 34.88g (+/- 2.05g, n=4); non-gravid females 34.70g (+/- 0.92g, n=13); gravid females 36.07g (+/- 0.43g, n=22).
GENETIC CHARACTERISTICS 2n=32. FN=60. All autosomes are biarmed. X chromosome is a medium-sized submetacentric, Y chromosome is a small acrocentric. Entire series of autosomal chromosomes grades smoothly in size from large to small. 2 rDNA sites and 2 teleometic chromosomes (Baker 1967). G and C-banding patterns identical to P.hastatus (Varella-Garcia et al 1989).
EXTERNAL MEASUREMENTS: A large bat but a small Phyllostomus.
Podtiaguin (1944) gives the following measurements for males (n=4) and females (n=2) in Paraguay: HB: male 70mm female 69-70mm; FA: male 56-57mm, female 56-58mm; TA: male 8mm, female 8mm; Thumb male 10mm female 10mm; First phalange male 14-16mm female 14-16mm; Second phalange male 28-29mm female 28mm; Third phalange male 15mm female 15mm.
Power & Tamsitt (1973) gave the following mean forearm measurements from across the species wide range (n=138 males, 85 females): FA: male 61.79mm, female 61.40mm.
Giannini & Brenes (2001) gave the following masses for unsexed adults (n=21) from Costa Rica: WT: 38.2g (+/- 3.1g). Valdivieso & Tamsitt (1962) give the following ranges of measurements for eight specimens from Colombia: HB: 69-92mm; TA: 12-18mm; FT: 12.1-15.1mm; FA: 57.5-64.1mm; EA: 19.4-23-5mm.
Anderson (1997) gives the following measurements for 6 males and 4 females from Bolivia: HB: male 85-95mm, female 88-89mm; TA: male 9-16mm, female 10-18mm; FT: male 10-20mm, female 15-20mm; FA: male 57-64mm, female 59-62mm; EA: male 15-21mm, female 19-22mm; WT: male 18-31g, female 31-32g.
SIMILAR SPECIES: This is a small Phyllostomus considerably smaller than P.hastatus the other Paraguayan species in all measurements. This species has a forearm in the range 56-58mm and a head and body length of 69-70mm, compared to forearm of 78-87mm and head and body of 95-115mm in hastatus. Note that the pelage does not extend onto the membranes, whereas in P.hastatus it extends onto the first third of the forearm and onto the uroptagium. Furthermore the tricoloured hairs of P.discolor give its pelage a grizzled effect, compared to the more uniform colouration of P.hastatus.
Structurally note that the calcar is shorter than the length of the hindfoot, whereas in all other members of the genus it is as long as or longer. Cranially the sagittal crest is reduced or absent. Dentally the first upper and lower incisors are wider than they are high, those of all other members of the genus are higher than they are wide.
DISTRIBUTION: In Bolivia the species has been recorded in Departamentos Beni, Cochabamba and Santa Cruz (Aguirre 2007). In Brazil the species has been recorded in the following states: Acre, Amazonas, Amapá, Bahía, Ceará, Distrito Federal, Espirito Santo, Minas Gerais, Mato Grosso do Sul, Mato Grosso, Pará, Paraiba, Pernambuco, Piauí, Paraná, Rio de Janeiro, Roraima and São Paulo (dos Reis et al 2007). The species is not known to occur in Argentina.
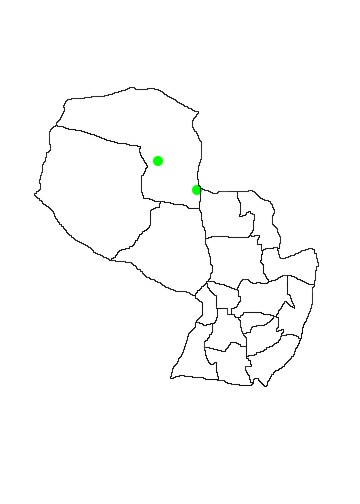
In Paraguay the species is known only from two localities in Departamento Alto Paraguay, Laguna Chamacoco near Fuerte Olimpo and Puerto Sastre (López-Gonzalez 2005). No specimens apparently exist today. Podtiaguin (1944) states that a specimen was collected by a G.Foster in 1901 at Puerto Sastre and sent to Dr Adolfo Pira in Stockholm, Sweden who initially identified it erroneously as a subadult male Phyllostomus hastatus. At the time of Podtiaguin´s publication a male and a female were supposedly deposited in the Museo de Historia Natural del Paraguay having been collected on 23 January 1914 at Laguna Chamococo 33 leagues west of Fuerte Olimpo and given to the museum by Sr. Domingo Gallea. Podtiaguin himself claims to have seen three individuals captured at Puerto Sastre by RPJ Méndez Baya in 1939 and stored in Baya´s magnificent collection. Podtiaguin provides a detailled description of the specimens that he saw as well as measurements and a summary of the characteristics that distinguish them from Phyllostomus hastatus, the only confusion species.
HABITAT: A habitat generalist able to utilise a range of both humid and dry habitats when sufficient food resources and roost sites are available. Willig et al (2007) found the species exclusively in human-altered agricultural areas in the Iquitos region of Peru.
In Brazil the species occurs in primary and secondary forest but is also found in heavily-altered environments such as villages and banana plantations (dos Reis et al 2007). Willig (1985) found the species to be rare to common in the caatinga of northeast Brazil but abundant in cerrado. In Bolivia the species has also been recorded in flooded grasslands and exceptionally humid foothill forest (Aguirre 2007).
In Paraguay the two cited locations are in chaco habitat, one in typical dry thorn forest and the other in humid riverine semideciduous forest (López-Gonzalez 2005).
ALIMENTATION: Willig (1985) considers the species an omnivore.
Foraging Behaviour and Diet Aguiar & Marinho-Filho (2007) identified Coleoptera and Hymenoptera in fecal pellets of a single individual from this species in Atlantic Forest in Minais Gerais, Brazil.
Diet in Captivity The first Colombian specimens were captured in nets near vegetation with fruiting papaya, banana, guayaba and almonds (Valdivieso & Tamsitt 1962). Goodwin & Greenhall (1981) note that the species was maintained in captivity in Trinidad on a diet of soft fruit such as ripe bananas, mangos and pawpaws but that it rejected meat. The long, extensile tongue is deeply-grooved on the upper surface and this is used to scoop out fruit pulp.
Giannini & Brenes (2001) studied the flower-feeding behaviour of this species under captive conditions in Costa Rica. Presented with banana inflorescences, the bats approached in direct flight, rotated in the air and landed head downwards. They probed a single flower for about a second before flying away and returning shortly afterwards, repeating this behaviour on numerous occasions (mean = 16 +/-10). Constant revisiting of the flower was suggested as a behavioural adaptation to allow this heavy bat to visit delicate flowers aimed more at small, hovering nectarivorous bats and unable to withstand the weight of a larger bat perching on it - representing an intermediate tactic between hovering and larger landing bats. Total mean foraging time was 181 seconds (+/-83s) with the reaction time from being presented with the flowers 320 seconds (+/-123s). Flowers were usually approached from the side or below, and only once was a flower approached from above, the bat then crawling head downwards to the flower. A mean of 74% (range 39-96%) of the nectar supply was consumed and foraging efficiency (mean microlitres of consumed nectare per gram of mean body mass) was 30.
REPRODUCTIVE BIOLOGY: Harem breeders, a single male maintaining a group females. The species is bimodally polyoestrous. No breeding data specificially for Paraguay is available.
Seasonality In southern Brazil breeding coincides with the wettest months of the year (dos Reis 2007). Willig (1985) notes a pregnant, lactating female from the caatinga of northeast Brazil in March and lactating, non-pregnant females in March, October and November. In the cerrado pregnant females were recorded in January (2, both lactating), February (2, none lactating), March (1 lactating), April (7 all lactating), September (4, 1 lactating), October (1 lactating) and December (16, 9 lactating). Lactating, non-pregnant females were recorded in March (1), April (2), September (2), October (5) and December (1), whilst females showing no signs of reproductive activity were reported for January (1), February (2), March (2), September (1), October (1) and December (1).
Pregnant females were captured in Colombia during the rainy season in October, but bats caught in the same area during the dry season in June were not pregnant (Valdivieso & Tamsitt 1962). Goodwin & Greenhall (1981) give the following data for Trinidad: gravid females in February, March, June and August; lactating females in August, September and October and males in breeding condition in January, August and October. Carter et al (1981) note that non of the ten females they collected in Trinidad from 2 to 8 August were pregnant. In Venezuela 1 of 4 specimens captured in April was lactating (August & Baker 1982). Hice et al (2004) found one lactating and one non-reproductive female in northeast Peru during June.
Courtship Typically males maintain an annual harem of 12 to 20 females, though harem composition is unstable, with females often moving between groups (dos Reis et al 2007).
GENERAL BEHAVIOUR: Unknown in Paraguay, though this is a widespread and often common bat in other areas.
Activity Levels Aguirre (2007) notes that the species is most active in savannas in Departamento Beni, Bolivia between 8 and 10pm.
Roosts Goodwin & Greenhall (1981) noted a preference for roosting in hollow Ceiba pentandra trees in Trinidad and that colonies of up to 25 individuals of both sexes form. Typically they roosted high up and well away from the single entrance. In Trinidad the species has been recorded as roosting in association with Saccopteryx bilineata, Noctilio leporinus and Carollia perspicillata of the species of confirmed or potential occurrence in Paraguay. Podtiaguin (1944) notes that three Paraguayan specimens collected by Méndez Baya were found hanging under a palm leaf but that they more typically prefer hollow trees.
Valdivieso & Tamsitt (1964) speculated that the strong-smelling, milky secretions of the chest gland may act to facilitate individual recognition and to advertise day roosts to conspecifics.
Flight Pattern Giannini & Brenes (2001) gave the following measurements associated with flight for unsexed adults (n=21) from Costa Rica: Wing loading 13.6Nm2 (+/- 1.22Nm2); Aspect ratio (winspan/area of wings and chest) 7 (+/- 0.25); Tip area index (area of dactylopatagium or wing tip area between the fingers/area of wing) 0.8 (+/- 0.95); Tip length index (length of Digit III/length of arm and forearm 1.36 (+/- 0.04); Tip shape index (tip area index/tiplength index - tip area index) 1.5 (+/- 0.27).
Grooming Behaviour Seven of nine females collected on Trinidad in August were moulting (Carter et al 1981).
Parasites Goodwin & Greenhall (1981) list the following parasites of this species in Trinidad: Trombicula carmenae (Trombiculidae), Euctenodes mirabilis, Trichobius costalimai, T.mixtus, T.perspicillatus (Streblidae) and unidentified trematodes.
VOCALISATIONS: Juveniles immediately give isolation calls when separated from their mother and can modify these to match the maternal directive call they hear from their mother (Esser 1994).
HUMAN IMPACT: None in Paraguay where it is a little known species with an apparently restricted range. Elsewhere this species is common, large and conspicuous, and frequently found in areas of human habitation.
CONSERVATION STATUS: Globally considered to be of Low Risk Least Concern by the IUCN, see http://www.iucnredlist.org/details/17216/0 for the latest assessment of the species. This species is under no immediate threat over most of its range and may actually benefit from habitat fragmentation in some areas. Willig et al (2007) for example found the species exclusively in human-altered agricultural areas in the Iquitos region of Peru and absent from pristine habitats nearby. Paraguay represents the southern limits of the species range. The lack of recent records from Paraguay are likely a product of the isolated nature of the species range and it should be considered Data Deficient at national level.
CITE AS: Smith P (2009) FAUNA Paraguay Online Handbook of Paraguayan Fauna Mammal Species Account 37 Phyllostomus discolor.
LAST UPDATED: 20 August 2009.
REFERENCES:
Aguiar LMS, Marinho-Filho J 2007 - Bat Frugivory in a Remnantof Southeastern Brazilian Atlantic Forest - Acta Chiropterologica 9: p251-260.
Aguirre LF 2007 - Historia Natural, Distribución y Conservación de los Murciélagos de Bolivia - Fundación Simón I. Patiño, Santa Cruz.
Allen JA 1904 - List of Mammals from Venezuela Collected by Mr Samuel M Klages - Bulletin AMNH 20: P337-345.
Anderson S 1997 - Mammals of Bolivia Taxonomy and Distribution - Bulletin AMNH 231.
August PV, Baker RJ 1982 - Observations on the Reproductive Ecology of Some Neotropical Bats - Mammalia 46: p177-181.
Baker RJ 1969 - Karyotypes of Bats of the Family Phyllostomidae and Their Taxonomic Implications - Southwestern Naturalist 12: p407-428.
Braun JK, Mares MA 1995 - The Mammals of Argentina: An Etymology - Mastozoologia Neotropical 2: p173-206.
Carter CH, Genoways HH, Loregnard RS, Baker RJ 1981 - Observations on Bats from Trinidad with a Checklist of Species Occurring on the Island - Occasional Papers of the Museum of Texas Tech University 72.
Gardner AL 2007 - Mammals of South America Vol 1: Marsupials, Xenarthrans, Bats and Shrews - University of Chicago Press, Chicago.
Gervais P 1856 - Deuxième Mémoire. Documents Zoologiques pour Servir a la Monographie des Chéiroptères Sid-Américains p25-88 in de Castelnau F, Animaux Nouveaux our Rares Recueillis Pendant l´Expédition dans les Parties Centrales de l´Amérique du Sud, de Rio de Janeiro a Lima et de Lima au Para, Exécutée par Ordre du Governement Français Pendant les Années 1843 a 1847 su la Direction de Comte Francis de Castelnau - Bertrand, Paris.
Elliot DG 1905 - Descriptions of Apparently New Species and Subspecies of Mammals from Mexico and San Domingo - Proceedings of Biological Society of Washington 18: p233-236.
Emmons LH 1999 - Mamíferos de los Bosques Húmedos de América Tropical - Editorial FAN, Santa Cruz.
Esser KH 1994 - Audio-vocal Learning in a Non-human Mammal: The Lesser Spear-nosed Bat Phyllostomus discolor - Neuro Report 5: p1718-1720.
Giannini NP, Brenes FV 2001 - Flight Cage Observations of Foraging Mode in Phyllostomus discolor, P.hastatus and Glossophaga commissarisi - Biotropica 33: p536-550.
Goodwin GG, Greenhall AM 1961 - A Review of the Bats of Trinidad and Tobago: Descriptions, Rabies Infection and Ecology - Bulletin AMNH 122: p187-302.
Hice CL, Velazco PN, Willig MR 2004 - Bats of the Reserva Nacional Allpayuaho-Mishana, Northeast Peru, with Notes on Community Structure - Acta Chiropterologica 6: p319-334.
López-Gonzalez C 2005 - Murciélagos del Paraguay - Biosfera Numero 9.
Miller GS 1907 - The Families and Genera of Bats - Bulletin USNM 57.
Podtiaguin B 1944 - Contribuciones al Conocimiento de los Murciélagos del Paraguay - Revista de la Sociedad Cientifica del Paraguay 6: p25-62.
Power DM, Tamsitt JM 1973 - Variation in Phyllostomus discolor (Chiroptera: Phyllostomatidae) - Canadian Journal of Zoology 51: p461-468.
Redford KH, Eisenberg JF 1992 - Mammals of the Neotropics: Volume 2 The Southern Cone - University of Chicago Press, Chicago.
Reis dos NR, Peracchi AL, Pedro WA, Lima de IP 2007 - Morcegos do Brasil - Londrina, Brazil.
Santos M, Aguirre LF, Vázquez LB, Ortega J 2003 - Phyllostomus hastatus - Mammalian Species 722: p1-6.
Taddei VA 1975 - Phyllostomidae (Chiroptera) do Norte-Occidental do Estado de São Paulo I: Phyllostominae - Ciencia e Cultura 27: p621-632.
Tschudi JJ von 1844 - Untersuchungen Über die Fauna Peruana - Scheitlin und Zollikoffer, St. Gallen.
Valdez R 1970 - Taxonomy and Geographic Variation of the Bats of the Genus Phyllostomus - Texas A&M University, PhD Dissertation.
Valdivieso D, Tamsitt JR 1962 - First Records of the Pale Spear-nosed Bat in Colombia - Journal of Mammalogy 43: p422-423.
Valdivieso D, Tamsitt JR 1964 - Histology of the Chest Gland of the Pale Spear-nosed Bat - Journal of Mammalogy 45: p536-539.
Varella-García M, Morielle-Versute E, Taddei VA 1989 - A Survey of Cytogenetic Data on Brazilian Bats - Revista Brasileira de Genetica 12: p761-793.
Wagner JA 1843 - Diagnosen Neuer Arten Brasilischer Handflügler - Arch. Naturgesch. 9: p365-368.
Willig MR 1983 - Composition, Microgeographic Variation and Sexual Dimorphism in Caatinga and Cerrado Bat Communities from Northeast Brazil - Bulletin of the Carnegie Museum of Natural History 23: p1-131.
Willig MR 1985 - Reproductive Activity of Female Bats from Northeast Brazil - Bat Research News 26: p17-20.
Willig MR, Pressley SJ, Bloch CP, Hice CL, Yanoviak SP, Díaz MM, Chauca LA, Pacheco V, Weaver SC 2007 - Phyllostomid Bats of Lowland Amazonia: Effects of Habitat Alteration on Abundance - Biotropica 39: p737-746.
MAP 37:
Phyllostomus discolor


